Fluorobenzene
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name Fluorobenzene | |||
| Other names Phenyl fluoride Monofluorobenzene | |||
| Identifiers | |||
CAS Number |
| ||
3D model (JSmol) |
| ||
Beilstein Reference | 1236623 | ||
| ChEBI |
| ||
| ChEMBL |
| ||
| ChemSpider |
| ||
| ECHA InfoCard | 100.006.657 | ||
| EC Number |
| ||
Gmelin Reference | 49856 | ||
| KEGG |
| ||
PubChem CID |
| ||
| UNII |
| ||
| UN number | 2387 | ||
CompTox Dashboard (EPA) |
| ||
InChI
| |||
| |||
| Properties | |||
Chemical formula | C6H5F | ||
| Molar mass | 96.103 | ||
| Appearance | Colorless liquid | ||
| Density | 1.025 g/mL, liquid | ||
| Melting point | −44 °C (−47 °F; 229 K) | ||
| Boiling point | 84 to 85 °C (183 to 185 °F; 357 to 358 K) | ||
| low | |||
| -58.4·10−6 cm3/mol | |||
Refractive index (nD) | 1.46553 | ||
| Structure | |||
| Planar | |||
| Hazards | |||
| GHS labelling: | |||
   | |||
| Warning | |||
| H225, H318, H411 | |||
| P210, P233, P240, P241, P242, P243, P264, P273, P280, P303+P361+P353, P305+P351+P338, P310, P337+P313, P370+P378, P391, P403+P235, P501 | |||
| NFPA 704 (fire diamond) |  1 3 0 | ||
| Related compounds | |||
Related halobenzenes | Chlorobenzene Bromobenzene Iodobenzene | ||
Related compounds | Benzene 1,2-Difluorobenzene | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  Y verify (what is Y verify (what is  Y Y N ?) N ?) Infobox references | |||
Fluorobenzene is an aryl fluoride and the simplest of the fluorobenzenes, with the formula C6H5F, often abbreviated PhF. A colorless liquid, it is a precursor to many fluorophenyl compounds.
Preparation
PhF was first reported in 1886 by O. Wallach at the University of Bonn, who prepared the compound in two steps. Phenyldiazonium chloride was first converted to a triazene using piperidine:
- [PhN2]Cl + 2 (CH2)5NH → PhN=N-N(CH2)5 + [(CH2)5NH2]Cl
The triazine was then cleaved with hydrofluoric acid:
- PhN=N-N(CH2)5 + 2 HF → PhF + N2 + [(CH2)5NH2]F
Historical note: in Wallach's era, the element fluorine was symbolized with "Fl". Thus, his procedure is subtitled "Fluorbenzol, C6H5Fl".[1]
On the laboratory scale, PhF is prepared by the thermal decomposition of the benzenediazonium tetrafluoroborate:
- PhN2BF4 → PhF + BF3 + N2
According to the procedure, solid [PhN2]BF4 is heated with a flame to initiate an exothermic reaction, which also affords boron trifluoride and nitrogen gas. Product PhF and BF3 are readily separated because of their differing boiling points.[2]
The technical synthesis is by the reaction of cyclopentadiene with difluorocarbene. The initially formed cyclopropane undergoes a ring expansion and subsequent elimination of hydrogen fluoride.
Reactions
PhF behaves rather differently from other halobenzene derivatives owing to the pi-donor properties of fluoride. For example, the para position is more activated than benzene toward electrophiles. For this reason, it can be converted to 1-bromo-4-fluorobenzene with relatively high efficiency.[3]
Solvent properties
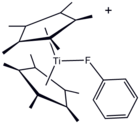
Structure of [(C5Me5)2Ti(FC6H5)]+, a coordination complex of fluorobenzene.
PhF is a useful solvent for highly reactive species. Its melting point at -44 °C is lower than that of benzene. In contrast, the boiling points of PhF and benzene are very similar, differing by only 4 °C. It is considerably more polar than benzene, with a dielectric constant of 5.42 compared to 2.28 for benzene at 298 K.[4] Fluorobenzene is a relatively inert compound reflecting the strength of the C–F bond.
Although it is usually considered a non-coordinating solvent, a metal complex of PhF has been crystallized.[5]
See also
References
- ^ Wallach, O. "Über einen Weg zur leichten Gewinnung organischer Fluorverbindungen" (Concerning a method for easily preparing organic fluorine compounds) Justus Liebig's Annalen der Chemie, 1886, Volume 235, p. 255–271; doi:10.1002/jlac.18862350303
- ^ Flood, D. T. (1933). "Fluorobenzene". Org. Synth. 13: 46. doi:10.15227/orgsyn.013.0046..
- ^ Rosenthal, Joel; Schuster, David I. (2003). "The Anomalous Reactivity of Fluorobenzene in Electrophilic Aromatic Substitution and Related Phenomena". J. Chem. Educ. 80 (6): 679. Bibcode:2003JChEd..80..679R. doi:10.1021/ed080p679.
- ^ Table of Dielectric Constants of Pure Liquids. National Bureau of Standards. 1951.
- ^ R.N. Perutz and T. Braun "Transition Metal-mediated C–F Bond Activation" Comprehensive Organometallic Chemistry III, 2007, Volume 1, p. 725–758; doi:10.1016/B0-08-045047-4/00028-5.
- v
- t
- e
| HF | He | |||||||||||||||||
| LiF | BeF2 | BF BF3 B2F4 | CF4 CxFy | NF3 N2F4 | OF OF2 O2F2 O2F | F− | Ne | |||||||||||
| NaF | MgF2 | AlF AlF3 | SiF4 | P2F4 PF3 PF5 | S2F2 SF2 S2F4 SF4 S2F10 SF6 | ClF ClF3 ClF5 | HArF ArF2 | |||||||||||
| KF | CaF2 | ScF3 | TiF3 TiF4 | VF2 VF3 VF4 VF5 | CrF2 CrF3 CrF4 CrF5 CrF6 | MnF2 MnF3 MnF4 | FeF2 FeF3 | CoF2 CoF3 | NiF2 NiF3 | CuF CuF2 | ZnF2 | GaF3 | GeF4 | AsF3 AsF5 | SeF4 SeF6 | BrF BrF3 BrF5 | KrF2 KrF4 KrF6 | |
| RbF | SrF2 | YF3 | ZrF4 | NbF4 NbF5 | MoF4 MoF5 MoF6 | TcF6 | RuF3 RuF4 RuF5 RuF6 | RhF3 RhF5 RhF6 | PdF2 Pd[PdF6] PdF4 PdF6 | AgF AgF2 AgF3 Ag2F | CdF2 | InF3 | SnF2 SnF4 | SbF3 SbF5 | TeF4 TeF6 | IF IF3 IF5 IF7 | XeF2 XeF4 XeF6 XeF8 | |
| CsF | BaF2 | * | LuF3 | HfF4 | TaF5 | WF4 WF6 | ReF6 ReF7 | OsF4 OsF5 OsF6 OsF 7 OsF8 | IrF3 IrF5 IrF6 | PtF2 Pt[PtF6] PtF4 PtF5 PtF6 | AuF AuF3 Au2F10 AuF5·F2 | HgF2 Hg2F2 HgF4 | TlF TlF3 | PbF2 PbF4 | BiF3 BiF5 | PoF4 PoF6 | At | RnF2 RnF6 |
| Fr | RaF2 | ** | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
| ↓ | ||||||||||||||||||
| * | LaF3 | CeF3 CeF4 | PrF3 PrF4 | NdF3 | PmF3 | SmF2 SmF3 | EuF2 EuF3 | GdF3 | TbF3 TbF4 | DyF3 | HoF3 | ErF3 | TmF2 TmF3 | YbF2 YbF3 | ||||
| ** | AcF3 | ThF4 | PaF4 PaF5 | UF3 UF4 UF5 UF6 | NpF3 NpF4 NpF5 NpF6 | PuF3 PuF4 PuF5 PuF6 | AmF3 AmF4 AmF6 | CmF3 | Bk | Cf | Es | Fm | Md | No | ||||
- AgPF6
- KAsF6
- LiAsF6
- NaAsF6
- HPF6
- HSbF6
- NH4PF6
- KPF6
- KSbF6
- LiPF6
- NaPF6
- NaSbF6
- TlPF6
- Cs2AlF5
- Li3AlF6
- K3AlF6
- Na3AlF6
and pseudohalogenides
- BaSiF6
- BaGeF6
- (NH4)2SiF6
- Na2[SiF6]
- K2[SiF6]
- Li2GeF6
- Li2SiF6
- CBrF3
- CBr2F2
- CBr3F
- CClF3
- CCl2F2
- CCl3F
- CF2O
- CF3I
- CHF3
- CH2F2
- CH3F
- C2Cl3F3
- C2H3F
- C6H5F
- C7H5F3
- C15F33N
- C3H5F
- C6H11F
lanthanide, actinide, ammonium
- VOF3
- CrOF4
- CrF2O2
- NH4F
- (NH4)2ZrF6
- CsXeF7
- Li2TiF6
- Li2ZrF6
- K2TiF6
- Rb2TiF6
- Na2TiF6
- Na2ZrF6
- K2NbF7
- K2TaF7
- K2ZrF6
- UO2F2
- FNO
- FNO2
- FNO3
- KHF2
- NaHF2
- NH4HF2
and iodosyl
- F2OS
- F3OP
- PSF3
- IOF3
- IO3F
- IOF5
- IO2F
- IO2F3